Modern Physics
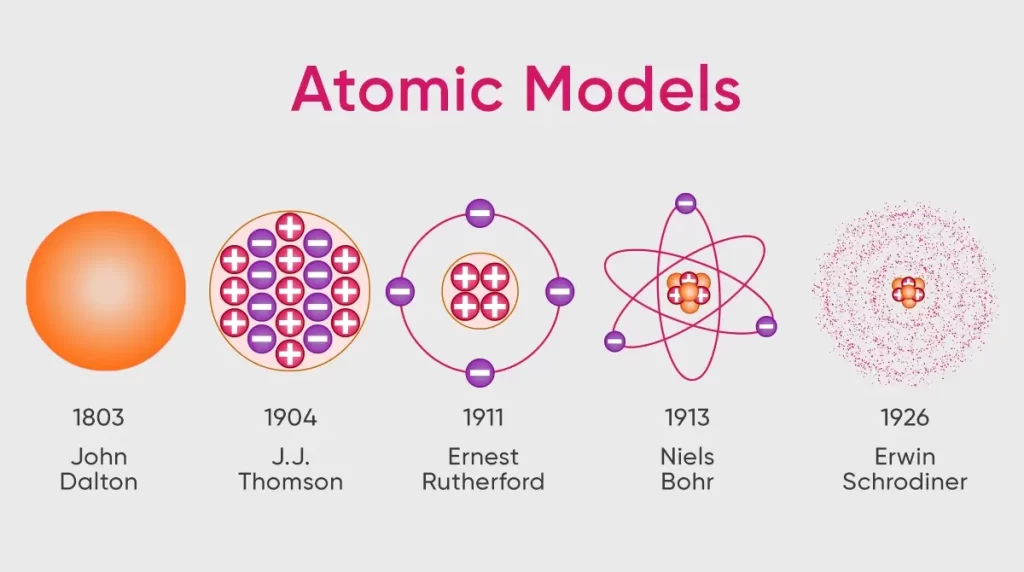
Model of an atom
| Protons | Discovered by Goldstein | Neutrons > Protons > Electrons (in terms of mass) |
| Electrons | Discovered by Thomson | |
| Neutrons | Discovered by Chadwick |
Radioactivity
- When size of nucleus enlarges > Electrostatic force > Nuclear force, which leads to radioactivity
- Unstable atomic nuclei will spontaneously decompose to form nuclei with a higher stability.
- The decomposition process is called radioactivity. The energy and particles which are released during the decomposition process are called radiation.
- When the unstable nuclei are prepared in the laboratory, the decomposition is called induced radioactivity.
- When unstable nuclei decompose in nature, the process is referred to as natural radioactivity.
Three major types of natural radioactivity
Alpha radiation
- Consists of a stream of positively charged particles, called alpha particles
- Alpha particles have an atomic mass of 4 and a charge of +2 (a helium nucleus).
- When an alpha particle is ejected from a nucleus, the mass number of the nucleus decreases by four units and the atomic number decreases by two units. For example:
23892U → 42He + 23490Th (The helium nucleus is the alpha particle)
Beta Radiation
- Consists of a stream of electrons, called beta particles.
- When a beta particle is ejected, a neutron in the nucleus is converted to a proton, so the mass number of the nucleus is unchanged, but the atomic number increases by one unit. For example:
23490Th → 0-1e + 23491Pa (The electron is the beta particle)
Gamma Radiation
- Gamma rays are high-energy photons with a very short wavelength (0.0005 to 0.1 nm).
- The emission of gamma radiation results from an energy change within the atomic nucleus.
- Gamma emission changes neither the atomic number nor the atomic mass.
- Alpha and beta emission are often accompanied by gamma emission, as an excited nucleus drops to a lower and more stable energy state.
Applications of Radioactivity
- Used as a tracer for chemical reactions. You can put an isotope in a living organism and it will do the same reactions as the regular element but you will be able to trace what it reacts with and where it goes
- Detecting how old something is by seeing how much of the isotope of the element is left > Carbon Dating > C 14 (Used for living organisms) & Uranium dating > For non-living organism ex. rocks
- Used for finding out the faults in metal structures esp. in airplanes > radioactive material will penetrate more through the cracked areas
- Act as a fuel for nuclear reactors to produce electricity
- Some isotopes are used in the treatment of cancer > to kill the cancer mutated cells
- Some isotopes are used to study the proper functioning of internal organs
- Gamma radiations are used to sterilize the surgical instruments
- Radio phosphorous is used for studying the rate of phosphorous assimilation by the plant
- Preservation of food grains and seeds
- Used for preparing synthetic elements (artificial transmutation)
- Detecting leaks in natural gas pipes
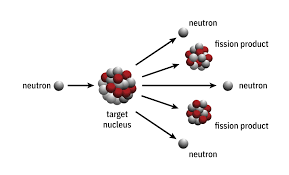
Nuclear fission
- In nuclear fission the nucleus of an atom breaks up into two lighter nuclei.
- The process is accompanied by the release of a large amount of energy.
- The process may take place spontaneously in some cases or may be induced by the excitation of the nucleus with a variety of particles (e.g., neutrons, protons, deuterons, or alpha particles) or with electromagnetic radiation in the form of gamma rays.
Atomic bomb > Only by fissile U 235 i.e. Enriched Uranium (90%)
For Nuclear reactors > 6 % of U-235
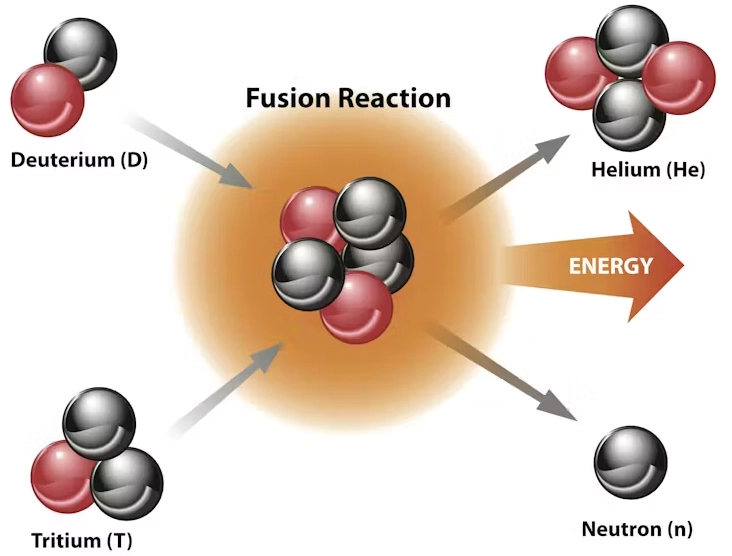
Nuclear fusion
- Process by which nuclear reactions between light elements form heavier elements (up to iron).
- During this process, matter is not conserved because some of the matter of the fusing nuclei is converted to photons (energy) > substantial amounts of energy are released.
Hydrogen bomb > Requires an atomic bomb to detonate
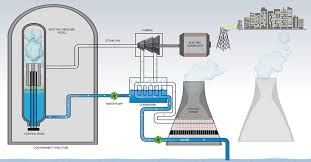
Nuclear Reactor
- A nuclear reactor, formerly known as atomic pile, is a device used to initiate and control a sustained nuclear chain reaction.
- Nuclear reactors are used at nuclear power plants for electricity generation and in propulsion of ships.
- Heat from nuclear fission is passed to a working fluid (water or gas), which runs through turbines.
- These either drive a ship’s propellers or turn electrical generators.
Sequential Process
| Fission | Controlled chain reaction of U 235 or Plutonium 239 |
| Moderators | D2O, H2O, Solid Graphite (To slow down neutrons bombardment & start a chain reaction) |
|
Heat Generation > Rotation of turbines > Powering Generator > Electricity through cable lines |
|
| Cooling | Liquid sodium |
| Control Rods | Cadmium (Which absorb excess neutrons) |
For more updates, explore the Physics. Feel free to share your thoughts and comments.
If you’re passionate about building a successful blogging website, check out this helpful guide at Coding Tag – How to Start a Successful Blog. It offers practical steps and expert tips to kickstart your blogging journey!