Electricity & Magnetism
Conductors & Charges
- When a hollow conductor is charged with static electricity, charge resides on outside surface of the conductor while inner surface remains unchanged.
- Hence, If a car is struck by lightning, person sitting inside are shielded from electricity & remain unharmed
- At absolute 0, metals almost reach 0 resistances & become superconductors. ex Hg
- Semiconductors > Electrical conductivity & resistivity is in-between conductors & insulators. For ex. Silicon & Germanium
- Resistance = Resistivity (length /area)
- Resistivity increases with increase in temp. in conductors but decrease with increase in temp. in semi-conductors
Electric cells
- Electrical Cell is a power generating device > Converts stored chemical energy into electrical energy.
- It is the combination of electrodes & electrolytes, where a difference of certain electric potential is established between the electrodes as a result of the chemical reaction between electrodes & electrolytes.
Based on packaging and shapes, Electrical Cells can can divided into following four categories
Based on the Electrical properties of the cell, Cells can can divided into following four categories
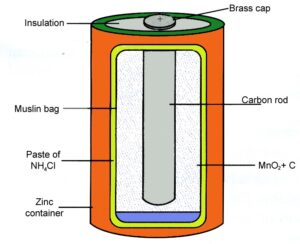
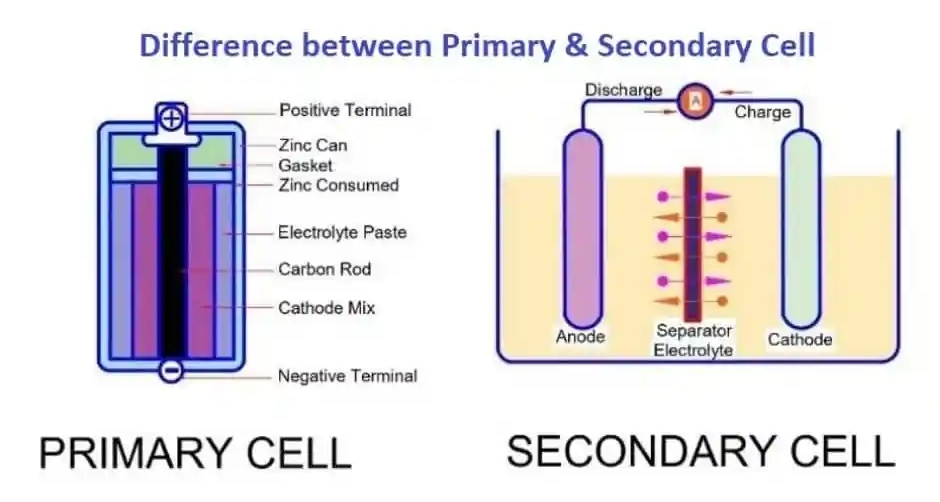
Primary Cell (Dry Cell)
- An Electrical cell which is powered by the irreversible chemical reaction is called Primary Cell.
- Since it is powered by irreversible chemical reaction, It cannot be recharged after being used
- Hence, it can be used only once and need to be disposed off after single use.
- Most of the primary cell contains their electrolytes in solid form absorbed within some absorbent material so these kind of cells are also called Dry Cell
- Nickel ion batteries, Lithium batteries zinc–carbon batteries and alkaline batteries etc.
Secondary Cell
- The Electrical Cell which can be electrically recharged after being used is called secondary cell.
- These cells are powered by reversible chemical reaction and the state of Electrodes & Electrolyte can be reversed to its original form by applying external power source after being used.
- Low resistance & provide large current > Lead cells used for ignition & lightening in cars
- Nickel-Cadmium, Nickel-Zinc, Nickel metal hydride, lithium-ion (Li-ion) cells, lead–acid battery
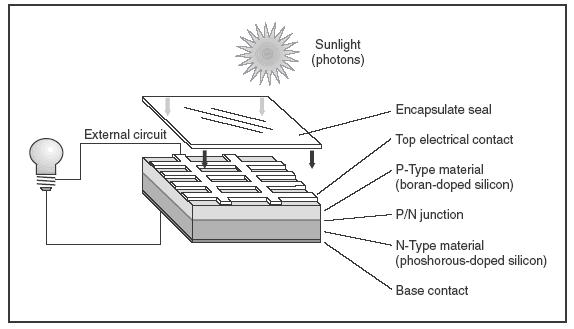
Reserve Cell
- Cells where one of the key components of the cell is separated from the rest of the components until activated manually or by some automatic means.
- These kinds of cell remain deactivated and nonfunctional until it is activated manually or by some means like heat, water or other means.
- One example is thermal cell (Photovoltaic) where the electrolyte remains inactive in its solid form until the heat applied melts the electrolyte to activate the cell.
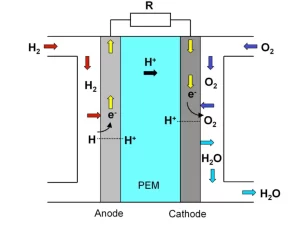
Fuel Cell
- A Fuel Cell is the kind of cell where the chemical energy from a fuel fed into the cell is converted into electrical energy through a chemical reaction with oxidizing agent.
- The fuel that is fed into a fuel cell can be Hydrogen, Hydrocarbons, Natural gas etc.
- A fuel cell can produce electricity as long as the fuel and the oxidizing agent are fed into the fuel cell.
Battery
- A single unit of electro-chemical generator is known as Electrical Cell, while the combination of several such units connected electrically is known as a Battery.
- Several cells are combined and connected electrically in series or parallel to form a battery which has two main terminal electrodes one Positive and one Negative.
- The electrical potential difference between the two main electrodes depends upon the numbers of cells, types of cells and the types of combination used to form the battery.
Some Basic terms used in electricity
| Dynamo / Electric Generator | Converts mechanical energy into electrical energy |
| Invertor | Converts DC > AC |
| Filament Lamp | Tungsten filament filled with argon gas (Highly heat producing > Wastage) |
| Fluorescent Tubes | Mercury vapours filled at low pressure (Lower heat producing) |
| Motor | Current carrying conductor when placed at right angle to the magnetic field, undergoes motion due to electromagnetic forceCoverts electric energy into mechanical energy |
| Fuse | A thin wire of tin-lead alloy with low melting pointNow days substituted by MCB – Miniature circuit board |
| 3 Pin Plug | Top Bigger Hole (Earth) — Earthed 1st before connecting to live circuitLeft Hole (Neutral) Right Hole (Live) |
| Cables | Red / Brown (Live wire); Blue / Black (Neutral); Green / Yellow (Earth) All the switches in house are put at live wire, if they were put in neutral wire, sockets would remain live even after switches are in off position & person will get sock |
For more updates about Electricity & Magnetism, explore the Physics. Feel free to share your thoughts and comments.
If you’re passionate about building a successful blogging website, check out this helpful guide at Coding Tag – How to Start a Successful Blog. It offers practical steps and expert tips to kickstart your blogging journey!