Heat & Thermodynamics
Thermodynamics
- (F – 32 / 180 = C/100) > At -40*C both scales show identical readings
- No upper limit of temp. is defined but have a definite lower limit i.e. absolute 0 k or -273*C
| 0*C = 237 K | 100*C = 373 k | Rise of temperature is equal in both the scales |
In cold countries we use alcohol in thermometer instead of mercury, because freezing point of mercury is -39*C & of alcohol is -115*C
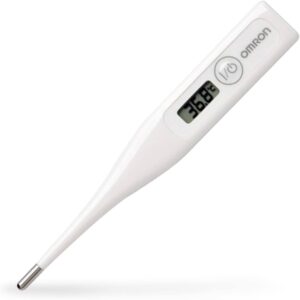
Electronic Thermometer
- Basic component thermistor, whose resistance changes with temperature
- Measures resistance & covert it to temp. as shown digitally
Water – Min Volume & Maximum Density at 4*C
- When temp. falls, top layer of water contracts & become denser & sinks to bottom
- This process goes on until whole water of the pond reaches 4*C i.e. at max density
- If temperature falls any further, top layer expands & remain at the top till it freezes and water under it remains at 4*C, leading to survival of aquatic life
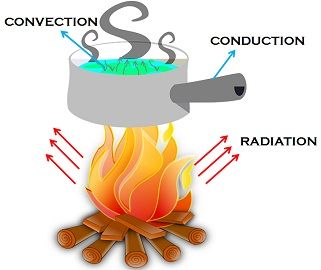
Heat Transfer Processes
Conduction
- Occurs in solids, when one end is heated then the other end also heats up
- Mainly due to transfer of heat though solids particles
Convection
- Occurs in liquids & gases, via circulation of particles to form convectional currents
- For ex. heating elements in geyser are fitted near bottom & cooling units in freeze are fitted near top
Radiation
- No medium is required (heating by electromagnetic waves)
- Good absorbers are good emitters too, for ex : Dark colors & rough surfaces
- Highly polished surfaces are poor radiators
Newton’s law of cooling
- Rate at which hot body loses heat is directly proportional to surrounding temperature.
- For ex. a body will come from 90*C to 80*C in less time then 40*C to 30*C
- Rate of cooling of hot water > rate of cooling of cold water in refrigerator
Cloudy nights are warmer than clear nights as clouds act as a blanket & reflect radiations emitted by earth back to the earth
Specific Heat Capacity
- Heat required to raise the temperature of a unit mass of substance by 1 k (unit joule/kg*kelvin)
- Specific heat of water > specific heat of land (1 cal = 4.2 j)
Water is used as a coolant in car engines mixed with ethylene glycol which acts as a lubricant, as a coolant & lowers the freezing point of water
Air conditioner
- Maintains temperature & humidity of a place
- 1 ton means transfer of 12000 BTU of heat from room in an hour
Boiling point of water vs atmospheric pressure
- Boiling point of water depends on external atmospheric pressure.
- When atmospheric pressure is at 76 cm of mercury, water boils at 100*C, but when external atm. pressure is increased, boiling point of water is also increased
- Increased boiling point allow water to hold more heat, which cooks the food faster
- At higher altitudes, atmospheric pressure is reduced, lowering the boiling point of water & food takes much longer to cook
Heat and Thermodynamics is the study of heat, energy, and their transformation. It involves laws of thermodynamics, heat transfer methods, energy conservation, entropy, and the behavior of systems in thermal equilibrium.
-
Explains heat transfer and temperature regulation.
-
Fundamental for engines, refrigerators, and heat pumps.
-
Vital in understanding the laws of energy conservation.
-
Used in meteorology and environmental science.
-
Important for material science and metallurgy.
-
Helps understand entropy and the direction of natural processes.
-
Essential in aerospace, chemical, and mechanical engineering.
- Explains climate change and greenhouse effect principles.
For more updates, explore the Physics. Feel free to share your thoughts and comments.
If you’re passionate about building a successful blogging website, check out this helpful guide at Coding Tag – How to Start a Successful Blog. It offers practical steps and expert tips to kickstart your blogging journey!









1 comment
hello sir , plz provide paper 2 IR portion notes.