Mixtures, Solutions & Chemical Processes
Mixtures are combinations of two or more substances that retain their individual properties. Solutions are homogeneous mixtures where solute dissolves in solvent.
Chemical Processes involve reactions leading to formation of new substances with different properties.
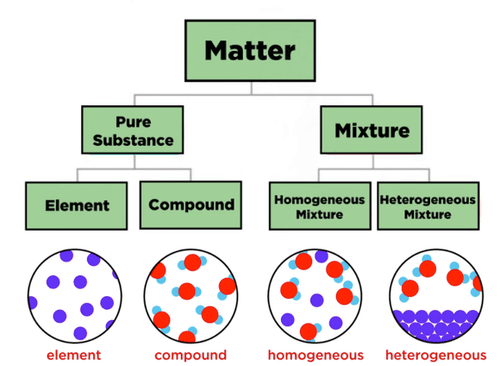
All matter can be broken down into two categories viz. pure substances and mixtures
- A pure substance is any matter that always has the same composition or “make up”.
- There are 2 classifications of pure substances: elements and compounds.
- An element is a substance that cannot be broken down into simpler substances. An element is made up of only one type of atom.
- A compound is a substance that is made up of 2 or simpler substances.
- A mixture is a combination of 2 or more substances “non-chemically”.
- This means we can separate a mixture without undergoing a chemical reaction or chemical change.
- All mixtures can be classified into 2 groups: homogeneous and heterogeneous.
- Homogeneous Mixture > (Solvent + Solute) not distinguished. for ex. solution & colloids
- Heterogeneous Mixture > (Solvent + Solute) readily distinguished. for ex. suspension mixture
Separation Methods of Mixtures
For 2 Solids
|
For 2 Liquids
|
| For Solids & Liquids |
|
Difference between Mixture & Compound
| Mixture | Compound |
|
|
Solution, Suspension & Colloids
Solution
- Homogeneous mixture with particle size < 1 nm diameter
- Does not separate out on keeping still & cannot be separated by filtration
- Does not scatter light due to extremely small particles
- Examples include: Carbonated drinks, Salt & Sugar solution
Colloids
- Heterogeneous but appears homogeneous with particle size b/w 1 – 100 nm in diameter
- Does not separate out on keeping still
- Separates out on centrifugation, not by filtration
- Scatters a beam of light – Tyndall Effect
- Include Milk, Blood, Soap solution, Ink, Jelly, Starch solutions, Body lotions, Foam, Gels, Gemstones etc.
Suspension
- Heterogeneous mixture with particle size > 100 nm in diameter
- Particles settle down on keeping still & separates out by filtration
- Scatters a beam of light with particles of the solute seen easily.
- Examples include: Sand in water, Milk of magnesia, Chalk solution in water
Solubility
- Maximum quantity of solute in 100 gm of solvent at a particular temperature
- Increases on increasing the temperature in liquids ( Decreases in case of gas)
- No effect of pressure in liquids (solubility increases in case of gases on increasing pressure)
- Saturated Solution > No more solute can be dissolved without increasing temperature
- Unsaturated Solution > More solute can be dissolved without increasing temperature
Important Chemical Processes
Diffusion
- Spreading & mixing of one substance with the another due to the motion of particles, which continues till a uniform mixture is formed
- Mixing particles move in zigzag known as Brownian motion
- Increases with temperature, fastest in gases & slowest in solids
- Few of the examples are : Dust particles in air, Smell of perfume in air, Spread of virus on sneezing, CO2 & O2 in water, Leakage of LPG (Identified by ethyl merceptane – a strong smelling substance)
Osmosis
- Diffusion is the case of using semi permeable membrane b/w 2 mixtures
- Only solvent is allowed to pass, from higher solvent conc. to the lower
- Few of the examples are:
- Preserving of pickle in salt
- Swelling up of raisins in water
- Earthworm dying when come in contact with salt
Dialysis
- Same as osmosis but for transplantation of kidney in humans
- Passes solvent & blocks waste
Latent Heat
- To change the state of a substance from one to heat energy is required.
- Latent heat does not increase temp. of a substance but is used in overcoming force of attraction b/w particle of a substance
It’s due to loss of latent heat that ice at 0*C is more effective in cooling than water at 0*C & due to gain of latent heat that steam at 100*C causes more severe burns than water at 100*C
Evaporation
- Transition of liquid to vapour at normal temperature
- Causes cooling when liquid evaporate as it draws latent heat of vaporisation from the liquid
- For ex. evaporation of ether or spirit from our hand, sweating, water kept in earthen pot
- Increases with temp, large surface area, wind speed & low humidity
Sublimation
- Transition of solids directly into gases without going through liquid state
- Ex: Dry ice, Camphor, Ammonium chloride, Iodine, Naphthalene, Anthracene
For more updates, explore the Physics. Feel free to share your thoughts and comments.
If you’re passionate about building a successful blogging website, check out this helpful guide at Coding Tag – How to Start a Successful Blog. It offers practical steps and expert tips to kickstart your blogging journey!