Atoms and Molecules
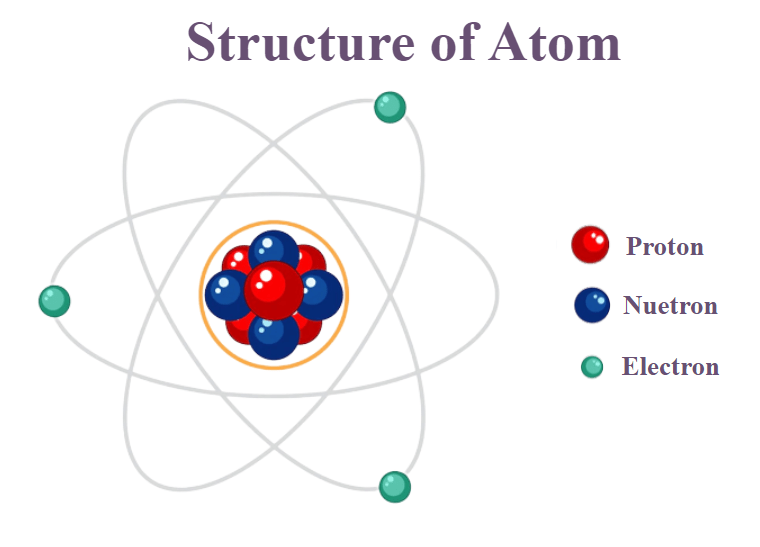
Structure of an Atom
- Matter has mass and takes up space. Atoms are basic building blocks of matter, and cannot be chemically subdivided by ordinary means.
- Atoms are composed of three types of particles viz. protons, neutrons, and electron.
- Mainly protons and neutrons are responsible for most of the atomic mass
- The nucleus of an atom consists of protons (each with a positive charge +1) and neutrons (zero charge).
- The electrons orbit round the nucleus, each with a negative charge of -1.
- A stable atom has the same number of electrons orbiting as it does protons in the nucleus; this makes the atom electrically neutral.
| Name | Charge | Location | Mass | Discovery |
| Proton | +1 | Atomic nucleus | 1.6726 X 10-27 kg | Goldstein |
| Neutron | 0 | Atomic nucleus | 1.6750 X 10-27 kg | Chadwick |
| Electron | -1 | Electron orbital | 9.1095 X 10-31 kg | Thomson |
- Atomic Number (Z) > Number of Protons (or of electrons) in an atom of an element
- Mass Number (A) > {Number of Protons (or Electrons) + Number of Neutrons} in an atom of an element
- Atomic or Molecular Mass > of times one atom or molecule of substance is heavier than 1/12 of carbon atoms
- Mass of 1 ‘C’ atom = 12 u where u = 1.66 * 10^ -24
- No. of electrons outer shells – 2n^2
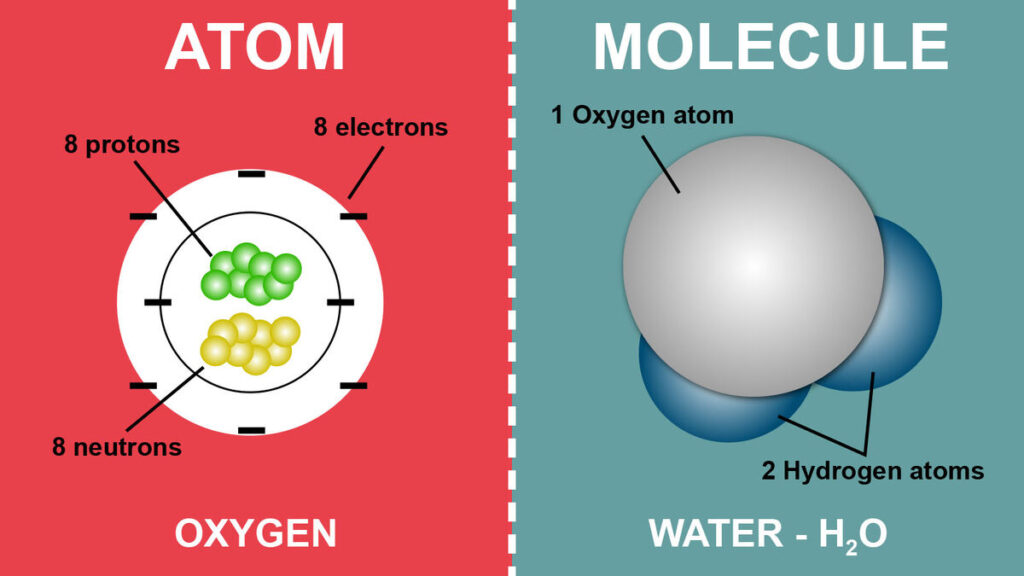
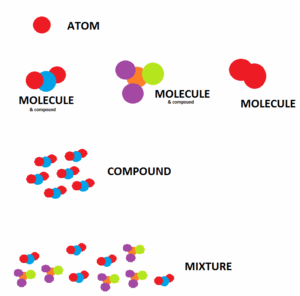
Elements, Molecules & Compounds
Element
- It is a substance that cannot be further resolved into simpler substances by chemical means.
- Itis a substance that is made entirely from one type of atom.
- For ex. Hydrogen, Carbon, Gold etc.
Compound
- It is a substance made of more than one type of atoms.
- It is a substance made from two or more different elements that have been chemically joined
- For ex. H2O), NaCl, CaCO3
Molecules
- This is the smallest unit of a compound.
- A molecule is formed when two or moreatoms join together chemically.
- All compounds are molecules but not all molecules are compounds.
- Molecularhydrogen (H2), molecular oxygen (O2) and molecular nitrogen (N2) are not compounds because each is composed of a single element.
- Water (H2O), carbon dioxide (CO2) and methane (CH4) are compounds because each is made from more than one element
Valence Electrons
Atoms that are missing an electron (that is, have one more proton than electrons) will tend to attract additional electrons, while atoms that have an excess electron (one more electron than protons) will tend to eject the outer-most electron.
- The shell containing the outer-most electron is called the valence shell, and is the chemically active shell.
- Electrons present in outer shell, taking part in a chemical reaction to fulfill outermost shell are known as valence electrons
- Valency > Number of valence electrons required or donated to fulfill outermost shell
| Covalency |
|
| Electrovalency |
|
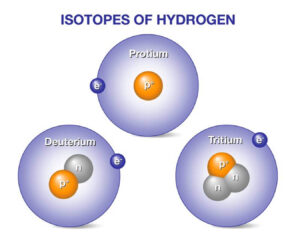
Isotopes
- Atoms with the same number of number of protons & electrons but different number of neutrons
- Hence, same atomic number but different atomic mass
- By changing the number of neutrons, isotopes still maintain the same overall neutrality and hence the chemical behavior remains unchange
- Hence same chemical properties but different physical properties
- Examples include – Protium (1H), Deutirium (2H) & Tritium (3H)
- Tritium is used in thermonuclear devices + also acts as a tracer
Radioactive decay
- Some isotopes are unstable, especially those with a lot of neutrons compare with the number of protons in the nucleus.
- These isotopes tend to eject some particles, in the form ofradiation, until a stable nucleus is produced.
- Such ejection process is called theradioactive decay.
- Isotopes that undergo radioactive decay are calledradioisotopes or radionuclides.
Radioactive Isotopes |
|
| Uranium Dating – For non-carbon containing materials | Serves as fuel for nuclear reactors for Ex. U -235 |
| Radiodating – C 14 (For carbon containing materials) | Goiter Treatment – Radiaoactive Iodine |
| Nuclear Bombs – U 235 & Plutonium 239 | To Detect Tumors – Arsenic 74 |
| To detect blood clot – Sodium 24 | Cancer treatment (Radiotherapy) – Co 60, Radon |
Half life
- A unit used to measure the stability of a radioisotope
- It is the time taken for half of the radioisotope in a sample to undergo radioactive decay
- Stable radioisotopes may take years to decay while the unstable ones may disappear in fractions of a second
Isobars
- Same mass number but different atomic number
- Different number of protons (or electrons) but same number of neutrons + protons in the nucleus
- Examples > 40Ar18 & 40Ca20
Isodiapheres
- Same difference between (Neutrons – Protons) b/w two elements
For more updates, explore the Sci Tech. Feel free to share your thoughts and comments.
If you’re passionate about building a successful blogging website, check out this helpful guide at Coding Tag – How to Start a Successful Blog. It offers practical steps and expert tips to kickstart your blogging journey!







1 comment
very nice post