Acid Rain & Ozone Depleting Substances
Acid Rain
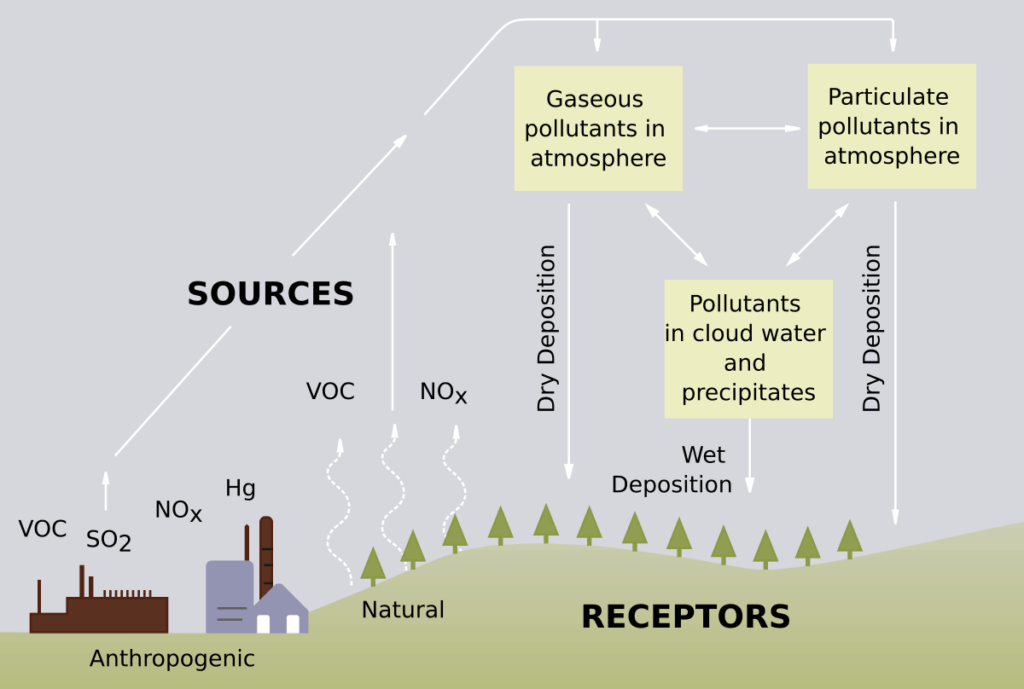
Acid rain (Ph < 5.6) is caused by emissions of sulfur dioxide and nitrogen oxide, which react with the water molecules in the atmosphere to produce acids (H2SO4& HNO3). Acid rain is a popular term referring to the deposition of wet (rain, snow, sleet, fog, cloud water, and dew) and dry (acidifying particles and gases) acidic components.
Ozone depleting substances (ODS)
- Scientific evidence indicates that stratospheric ozone is being destroyed by a group of manufactured chemicals, containing mainly chlorine or bromine, called “ozone-depleting substances” (ODS).
- ODS are very stable, nontoxic, non-flammable and environmentally safe in the lower atmosphere, which is why they became so popular in the first place
- However, their very stability allows them to float up, intact, to the stratosphere & once there, they are broken apart by the intense ultraviolet light, releasing chlorine and bromine.
- Chlorine and bromine demolish ozone at an alarming rate, by stripping an atom from the ozone molecule
- Main ODS – CFCs, HCFCs, Halons, CCl4, NOx, CH3CCl3 (Methyl chloroform)
- Major Sources: Solvent Cleansing products >Refrigeration & Air conditioners > Foam Products
Chlorofluorocarbons (CFCs)
- The most widely used ODS, accounting for over 80% of total stratospheric ozone depletion.
- Used as coolants in refrigerators, freezers and air conditioners in buildings and cars manufactured before 1995.
- Also found in industrial solvents, dry-cleaning agents and hospital sterilants.
- Also used in foam products — such as soft-foam padding (e.g. cushions and mattresses) and rigid foam (e.g. home insulation)
Also read: Ecological Succession, Food Chain, Food Web, Ecological Pyramids
Halons & Hydrobromofluorocarbon (HBFC)
- Halons, unlike CFCs, contain bromine, which destroys ozone 100 times more than chlorine
- Halons are used primarily in fire extinguishers & methyl bromide (Pesticide)
Methyl Chloroform
- Used mainly in industry as cleansing solvent — for vapour degreasing, cold cleaning, adhesives and chemical processing.
Carbon Tetrachloride
- Mainly used as solvent & in fire extinguishers.
Hydrofluorocarbons (HCFCs)
- Major, “transitional” substitutes for CFCs- much less harmful to stratospheric ozone than CFCs
- But still cause some ozone destruction and are potent greenhouse gases
Impacts of Ozone Depletion
- Skin cancers, sunburns and premature aging of the skin
- UV radiation can damage several parts of the eye, including the lens, cornea, retina and conjunctiva
- Weakening of the human immune system
- Several of the world’s major crop species are particularly vulnerable to increased UV, resulting in reduced growth, photosynthesis and flowering
- Plankton are threatened by increased UV radiation
Ozone hole mainly in Antarctica not in Arctic pole, because
- Southern polar region more colder & isolated than north, which favor the formation of polar stratospheric clouds (PSCs) which in turn serve as platforms for catalytic ozone breakdown
- As a consequence of having less land, the circulation over the Antarctic is more persistent and vortex-like, which favours PSCs
- Air inside the vortex is prevented from mixing with warmer, ozone-rich air from lower latitudes (This vortex is not a feature of the Arctic)
Ozone
- Ozone in atmosphere is measured in dobson unit.
- A Dobson unit of gas is equal to a layer of gas, at the surface of the Earth, with a thickness of one hundredth of a millimeter
- The ozone in the atmosphere is about 300 Dobsons, which means if you brought all of the ozone in the atmosphere down to a layer of pure ozone at the surface; it would be about three millimeters thick.
For more updates, explore the Environment category. Feel free to share your thoughts and comments
If you’re passionate about building a successful blogging website, check out this helpful guide at Coding Tag – How to Start a Successful Blog. It offers practical steps and expert tips to kickstart your blogging journey!











1 comment
very usefull notes , probide in hindi also if possible